Looking back several decades, chemists started paying closer attention to quaternary ammonium compounds. Tetrapropylammonium hydroxide (TPAOH) grew in importance during the latter half of the twentieth century, particularly as advanced material synthesis and catalysis shifted gears. Early research focused mostly on simpler, smaller molecular structures. As folks realized that ammonium species could direct the assembly of intricate frameworks like zeolites, TPAOH took center stage. Laboratories in Europe and the United States ramped up investigations, searching for bigger cations to help tune the porosity of new materials. Eventually, TPAOH found a permanent spot in the toolkit of scientists working on molecular sieves, layered silicates, and other advanced ceramics. That history set the stage for wide industrial and research use, cementing the substance’s reputation as a pivotal structural template and medium for complex reactions.
Tetrapropylammonium hydroxide goes by more than one name depending on who you ask. Sometimes it turns up as TPAOH, and not rarely, you’ll spot the alternative “quaternary ammonium base.” At its core, TPAOH consists of a central nitrogen atom surrounded by four propyl groups, plus a hydroxide anion tagging along. What that means for users comes down to a liquid that packs a strong punch in both organic and inorganic labs. Commercial suppliers usually offer it dissolved in water or small alcohols, with concentrations ranging anywhere from dilute to highly caustic. These solutions fill shelves in academic departments and specialty chemical warehouses, available for everything from zeolite synthesis to nucleophilic organic reactions.
TPAOH appears clear and colorless or faintly yellow in solution, though purity and storage can nudge this appearance a bit. With a sharp, unpleasant odor, its caustic bite becomes obvious on contact with skin or mucous membranes. In pure form, the compound does not form a solid easily under ambient conditions, preferring the comfort of room-temperature liquid state. The density of aqueous solutions depends on the exact concentration, usually hovering a bit above 1 gram per milliliter. Chemically, TPAOH acts as a strong base, splitting into its tetralkylammonium cation and hydroxide anion with ease. Folks experimenting in the lab notice that it handles moisture in the air poorly, picking up water and carbon dioxide, which slowly transforms the solution and clouds the quality unless tightly sealed.
Regulators and chemical suppliers emphasize detail when labeling TPAOH. Safety data sheets (SDS) always highlight its strong alkalinity and corrosive behavior. Labels list the chemical formula, often C12H29NO, and a CAS number that helps with recordkeeping and regulatory compliance. Standard bottles show concentration by weight and molarity, since researchers routinely require precise dosing to control framework assembly, synthesis yields, or pH adjustment. Labels warn of the need for gloves, splash-proof goggles, and proper ventilation, largely because exposure leads to burns or respiratory upset. Storage requirements hit the mark: cool, well-ventilated shelves, away from acids, oxidizers, and carbon dioxide sources.
To make TPAOH, chemists often start with tetrapropylammonium bromide. This salt undergoes a reaction with silver oxide in water, which encourages bromide ions to swap places with hydroxide ions. The reaction yields TPAOH and a byproduct—silver bromide, which drops out of the mixture, freeing up the new, strong base in the solution. After filtering and removing any solids, commercial producers may concentrate the liquid and check the precise strength before bottling. The entire process relies on careful measurement since slight mismatches in stoichiometry throw off purity and trample batch-to-batch consistency—a headache for high-stakes syntheses where reproducibility matters.
The robust base strength of TPAOH sees action on many fronts. Researchers turn to it for deprotonation, especially in cases where less aggressive bases can’t get the job done. Its structure, unlike small naked alkali hydroxides, offers some steric crowding; that lets chemists achieve selective reactions in both organic and inorganic chemistry. In the wild world of zeolite synthesis, TPAOH shapes silicon and aluminum atoms into tight, repeating cages. Modifications sometimes involve swapping the propyl moieties for other alkyl chains, tailoring the ammonium headgroup’s bulk or hydrophobicity for different roles—like ionic liquid precursors, intercalation agents, or phase transfer catalysts. Each tweak spins off its own set of possibilities, from synthesizing porous materials to mediating stubborn organic rearrangements.
Different catalogues and industries lean on their favorite terms. Techs and researchers call it TPAOH, tetrapropylammonium hydroxide, or by its IUPAC name: N,N,N-Tripropylpropan-1-aminium hydroxide. Occasionally, tetra-n-propylammonium hydroxide appears on product sheets, signaling the same beast dressed up in a new label. This diversity of synonyms shouldn’t get confused with naming conventions for less substituted ammonium bases; the propyl chains make a world of difference in how the molecule performs in solution and across applications.
Working with TPAOH doesn’t offer many shortcuts. The highly basic solution sears skin on contact, so trained personnel suit up with chemical-resistant gloves, lab coats, splash goggles, and sometimes face shields. Mechanical ventilation or fume hoods step in as insurance against accidental splashes or lingering fumes. Emergency showers and eyewash stations stand by if spills or splatters occur. On the environmental side, spills demand neutralization before disposal; mixing TPAOH with acids forms water and harmless salts, but releases heat and sometimes noxious fumes. Waste handling rules steer contaminated material toward licensed disposal services, curbing risks to water sources and plant operators. Regular audits, updated safety protocols, and routine training round out the essential safeguards wherever TPAOH gets stored, measured, or pipetted.
The calling card for TPAOH remains its starring role in zeolite synthesis. These complex, crystalline aluminosilicates anchor everything from gas separation membranes to industrial fluid catalytic cracking fields. TPAOH acts as both template and catalyst, shaping the microscopic structure of new zeolite types. Shifting out of materials science, researchers draw on the cation’s unique bulk for organic phase transfer, use in sol-gel science, and advanced surface chemistry studies. Electrochemical applications tap TPAOH for electrolyte development and precise pH regulation. In fields like analytical chemistry, the compound shows up in ion chromatography and specialized sample prep workflows. Intensive industries invest in this versatile base for ongoing projects in ceramics, glass, and multi-phase reactions.
Across research clusters worldwide, scientists push the limits of TPAOH’s utility. In molecular sieve chemistry, development teams tweak reaction pathways, exploring how small changes in base concentration or mixing speed affect the growth and morphology of zeolitic crystals. Surface scientists run trials with modified ammonium hydroxides, aiming to form new ion-exchange resins, advanced membranes, and hybrid organo-mineral materials. Some biotech labs eye the compound’s alkaline punch for cell lysis and nucleic acid isolation, while others monitor compatibility for sensor development or environmental monitoring. Funding agencies pour resources into greener, more efficient synthesis, less-toxic alternatives, and innovative process integration—a reflection of both the market’s appetite and the evolving sustainability imperative.
Moving beyond its physical dangers, the toxicology of TPAOH garners careful scrutiny. In vivo and in vitro experiments suggest that its strong alkalinity poses acute risk to mucous membranes, skin, and the respiratory tract. Swallowing or inhaling sizable amounts triggers corrosion, inflammation, and even systemic toxicity if the dose climbs high enough. Animal studies highlight organ-specific effects after repeated dose exposure, though most workplace guidelines revolve around proper handling and PPE, rather than chronic monitoring. Water systems contaminated with TPAOH show damage to aquatic life, especially under high exposure scenarios undiluted by natural mixing. Carefully published safety data and case studies from the chemical sector offer a clear lesson—nobody should underestimate this base or sidestep established safety routines when handling, transporting, or disposing of the substance.
Down the line, TPAOH stands to keep its value in both established and new technology sectors. As the market turns toward more sustainable and selective synthesis strategies, demand for specialized ammonium bases grows. Laboratory work on alternative “green” templates for advanced materials synthesis runs parallel to continued interest in traditional TPAOH, suggesting a dual path forward. In catalytic and separation sciences, incremental improvements in purity, handling, and safety may open up new applications—especially where tiny tweaks to structure or delivery translate into higher performance or fewer unwanted byproducts. Industrial and academic labs betting on electrification, renewable chemicals, and high-value ceramics continue to lean on TPAOH as a reliable workhorse, even as regulatory authorities push for safer packaging, tighter waste management, and cleaner downstream life cycles. Here lies a substance with roots in classic chemistry and branches that touch the frontiers of tomorrow’s science and technology.
Tetrapropylammonium hydroxide, or TPAOH for short, shows up in places you might not expect. It’s clear, strong-smelling, and mixes easily with water. Most people outside labs or factories rarely hear about it, but it has earned a reputation in chemical circles for one big reason: it opens the door to new materials and processes that power today’s technology.
The tech industry rides on nimble, complex materials. TPAOH plays an underrated role in shaping them. For example, silicon wafers need a little push to achieve the purity and patterning required in microchips. TPAOH enters the scene during the etching or polishing steps, ensuring the silicon looks and behaves just right. That’s not just good fortune—it’s deliberate chemical control helping manufacturers build smaller, more efficient devices.
From my own experience in research labs, I’ve watched chemists reach for TPAOH as they coax silicon into intricate designs. After the etching process, everyone in the room inspects patterns barely bigger than dust. When the liquid leaves sharp, clean features behind, folks breathe easy. In electronics, that precision adds real-world value to computers, phones, and everything in between.
Zeolites, the tiny framework crystals used in oil refineries and pollution control, depend on structure-directing agents during their birth in the lab. That’s where TPAOH steps in as a sort of chaperone. It guides the growing zeolite framework, acting much like a mold determines the shape of a cast statue. Without it, zeolites would lose their prized uniformity, and industries like petrochemicals and environmental science would feel the pinch.
Real-world numbers back this up. According to the American Chemical Society, zeolites command billions in global market value thanks to their use in catalysis, detergents, and air purification. TPAOH earns its place in this process, helping chemists deliver consistent structures batch after batch.
Working with TPAOH brings both promise and pitfalls. It’s caustic, stings on contact with skin, and puts lungs at risk if fumes spread. News stories occasionally highlight spills or poor storage, often linked to lax training. That’s not a failure of the chemical, but a sign companies and labs must keep safety top of mind.
Training goes a long way. Gloves, splash goggles, and fume hoods shouldn’t gather dust in any workplace using TPAOH. Labs and industry leaders owe it to their staff to foster a safety culture that includes clear labeling, emergency showers, and regular drills. I’ve seen firsthand how a bit of thought and preparation saves aggravation, health, and time down the line.
Some researchers look for ways to trim the need for strong bases like TPAOH or swap them out for greener choices. Companies test plant-derived solvents or reusable catalysts in search of similar performance with less environmental baggage. Funding for green chemistry projects continues to climb, and progress often comes from the front lines—techs and chemists brainstorming improvements before scaling up new processes. Every step in that direction chips away at the risks and can add up to a cleaner, safer industry.
Every time a name like tetrapropylammonium hydroxide lands in headlines, it starts conversations in labs, factories, and homes where people hear about chemicals but rarely know the full story. In my own experience in science media, most questions about chemicals come from children, parents, and workers who want to know—will this stuff hurt me? And should it be anywhere near my drinking water or workbench?
Tetrapropylammonium hydroxide finds its way into industrial and research settings more than kitchen cabinets. It’s popular with chemists who handle syntheses and with researchers making specialty materials. This isn’t the kind of compound that slips under the radar—it’s in safety datasheets, subject to agency review, and flagged if it escapes the technical world. The real concern? Hazards tied directly to how folks handle or store it.
This chemical packs a punch. It's a strong base, so touching the liquid can burn skin or eyes. Breathing the fumes irritates airways. If it splashes, reactions happen fast—redness, pain, maybe permanent injury if someone doesn’t rinse off right away. The worst accidents stem from skipping gloves, spilling near a face, or leaving an open lid in a small lab.
Authorities like the European Chemicals Agency and US EPA both flag tetrapropylammonium hydroxide as a corrosive and acutely toxic substance. After years of reviewing accident databases, it’s clear that mishandling corrosive bases causes more hospital runs than slow-acting toxins. It doesn't take much to turn a routine day into an emergency room visit.
Aside from burns, this compound poses real risks if swallowed or inhaled in concentrated form. It can damage tissues all the way down the throat and into the digestive system. Animal studies point out risks to organs after enough exposure, prompting researchers to recommend strict controls.
Opening chemical safety software drives the point home—tetrapropylammonium hydroxide doesn’t just create problems on contact. It can be toxic to aquatic life if released by accident. Even small spills can upset local water systems. Having spent time speaking with environmental teams, I know cleanup from a chemical spill isn’t always fast or easy, and every release creates headaches for entire communities.
Solutions exist, but they take buy-in from everyone—not just folks in lab coats. Simple steps like wearing chemical splash goggles and gloves stop most accidents. Every container should close tightly, labels need to stay visible, and emergency showers must stay in good working shape. Employers who train staff well and replace old equipment show they take these risks seriously.
Regulators deserve credit for enforcing strict rules on hazardous chemicals. Routes for reporting spills or exposures keep everyone accountable. But public awareness makes a difference too—local communities with easy access to safety guides and spill hotlines generally see fewer mishaps. In my own work, educational outreach turns dry instructions into habits that catch problems early.
Tetrapropylammonium hydroxide belongs in places where people know the risks and have the right tools and training. When handled with care and respect—it does its industrial job. Left unchecked, it can turn a simple process into a health crisis. People who ask about hazards are on the right track, and openness about risks helps everyone stay safer.
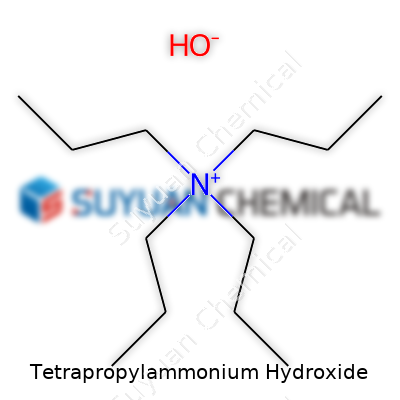
Tetrapropylammonium hydroxide carries the chemical formula C12H29NO. This formula shows twelve carbon atoms, twenty-nine hydrogens, one nitrogen, and one oxygen. Structurally, this compound features a nitrogen atom in the center, surrounded by four propyl groups and a hydroxide. In written shorthand, it looks like (C3H7)4N+ OH−.
Years spent in a research lab teach plenty about chemicals like this. Tetrapropylammonium hydroxide rarely ends up in the headlines, but it deserves attention—especially for its impact in chemical synthesis and biotechnology. It serves as a strong organic base, fitting into areas like organic synthesis, material science, and especially the production of zeolites. In fact, many modern zeolites owe their form and efficiency to this compound’s ability to act as a template, shaping the crystals for catalytic converters, water softeners, and environmental clean-ups.
During my graduate days, overlooking a single number in a chemical formula could topple an entire week’s work. Mistaking just one hydrogen or forgetting the charge on an atom often led research off the rails or wasted funding. In the case of tetrapropylammonium hydroxide, accurate formula use ensures researchers handle and store it safely and achieve the expected reactions every time. Without a clear formula, mixing or measuring gets risky. Some compounds use similar names and structures, so knowing exactly what’s in play proves critical not just for scientists, but also for anyone working on compliance or safety documentation.
Tetrapropylammonium hydroxide comes with its fair share of hazards. Touching it without gloves or breathing in its vapors can lead to skin burns, respiratory problems, or eye injuries. During my first industrial internship, a single drop splashed on a bench left a white residue that etched the surface, reminding us never to take strong bases lightly. So, each lab or plant must enforce gear and training, offering showers, masks, and fume hoods where this base is in use. Companies have been hit with fines for ignoring such guidelines, so keeping up with safety data sheets and emergency drills just makes sense.
Recognizing these health and practical concerns, a thoughtful approach to chemical handling pays off. Marking all containers with both the chemical name and formula reduces confusion, especially for new staff or visiting inspectors. In my experience, regular safety workshops that include hands-on demonstrations make a bigger difference than just sending around memos or guidelines.
Working with suppliers who provide full batch documentation and certificates of analysis ensures both lab teams and regulatory auditors know exactly what they’re using. That level of transparency doesn’t just boost safety; it also smooths collaboration between manufacturing, safety, and research. Stronger training, clear labeling, and reliable supply chains all reduce room for error.
Tetrapropylammonium hydroxide shows up often in industries that work with semiconductors or specialty chemicals. Most people in labs spot its chemical formula and reach for lab coats without a second look, but spending time with this compound reminds you just how vital smart handling is. I’ve seen the result of a single careless moment: broken safety gear, odors that stick in your memory, and reports filed to supervisors. There’s nothing routine about a strong base that reacts with moisture and attacks skin.
If you’ve ever cracked open a closed bottle of TPAH near a sink, you learn quick what happens when moisture joins the party. The solution degrades fast and fumes escape. I once watched someone lose almost half a container just because humidity in the air turned the top layer into a sticky mess. Every container worth using comes tightly sealed, and you want to keep it that way. Screw caps must fit tight. If a cap or seal wears out, replace rather than risk exposure.
Glass or high-density polyethylene stands up well to this material. I tried storing it in an off-brand plastic bottle once—big mistake. The sidewalls softened, then warped. That spilled and made a mess nobody forgot. Dedicated cabinets with ventilation help keep fumes from building up. Letting vapors collect means risking coughs or worse, so any spot that’s closed off or lacks airflow gets ruled out fast.
You don’t want TPAH warming up. Too much heat speeds up breakdown and can release more dangerous vapors. I remember leaving a container near a window on a cooler day, thinking nothing of it. Later, sunlight and radiant heat combined to make the lid hard to open, and the contents didn’t look the same. Always stash it in a cool, controlled environment, away from those sneaky heat sources and windows. If your space gets humid in the summer, it pays to run dehumidifiers or check for leaks.
It’s tempting to store similar bottles together, but mixing bases and acids spells trouble. I’ve worked in places where a mislabeled bottle meant hours of sorting and extra training. Label containers with big, clear text names and hazard symbols. Never put corrosives like TPAH next to acids, strong oxidizers, or anything flammable. People forget about shelf drift—over time, bottles migrate or new staff stack things without reading. Check shelves monthly to avoid surprises.
Safety data sheets often end up buried in files, and that’s a shame. Anyone handling, storing, or transporting this chemical ought to review the latest recommendations, from respirator options to emergency showers. Many shops run drills for spills, and I’ve seen that save injuries more than once. Eyewash stations within a short walk and thick gloves go from “nice idea” to “don’t work without them” after your first minor splash.
Absorbent pads and neutralizing agents stay stocked right under the nearest workspace. Everyone should feel comfortable grabbing them at the first sign of a leak. Reporting even minor mishaps means better safety for next time. The best crews I’ve worked with talk openly about close calls, because hiding mistakes leads to repeats. With chemicals like TPAH, the best outcome has always come from respect, not shortcuts.
Sometimes people get hung up on flashy lab breakthroughs, but real science relies on the everyday stuff, like Tetrapropylammonium Hydroxide. Plenty of industries, from semiconductors to organic synthesis, count on this chemical to keep production lines humming. Concentrations aren’t guesswork—they’re the foundation for safe, accurate, and efficient work. If you’ve ever handled a bottle of TPAH, you’ll know a label showing the right numbers actually means something.
You’ll find Tetrapropylammonium Hydroxide showing up most often as a solution—usually in water. The go-to choices typically land around 10%, 20%, and 25% by weight. These percentages aren’t picked out of thin air. People in labs want consistency, and these concentrations let manufacturers and researchers predict reactions with less hassle and better control.
On a basic level, a 25% solution feels like the workhorse for making zeolites or doing electronics cleaning. Go down to 10% or 20%, and you’re looking at options where you want less punch but still need action. A few suppliers will even put out solutions as high as 40% if anyone asks for it, but that gets tricky—higher concentrations ramp up the risk. It gets caustic quick, it absorbs moisture fast, and the bottle turns slick and hazardous before you know it. That’s not something you want near your hands if you’re the one measuring out the next beaker.
I remember adding a too-strong base by accident once. It only took one mistake to understand that a slightly off concentration turns a controlled reaction into a mess. If a solution’s too dilute, you’re wasting time waiting for things to happen. Too strong, and glassware or resin gets damaged, not to mention the risk of burns or ruined compounds.
Industries that rely on process control can’t afford those kinds of slip-ups. The semiconductor world expects results to the nanometer. Zeolite synthesis depends on careful timing and exact route-planning, especially since the wrong concentration derails the whole batch. Going with standard options, like 10%, 20%, or 25%, helps teams maintain a routine. There’s less chance of a misstep that costs money or hurts someone.
Sourcing TPAH straight from reputable chemical companies plays a big part in safety. These companies run a tight ship—they test solutions, lock down concentrations, and batch everything to match the label. I’ve seen plenty of homemade “adjustments” around research benches, and it always leads to second-guessing, double-checking, or worse, dangerous spills. Keeping things standardized cuts all that out.
Suppliers can help users by making data sheets easy to read. You don’t want to scroll through dozens of pages or guess at the pH or density. Simple charts showing product strength and safe handling go further than long explanations. Teams using TPAH daily want to open a new container and know instantly if they’re working with 10% or something stronger. Labels should scream out information, plain and simple.
Whether the bottle says 10% or 25%, you can’t forget about the basics—goggles, gloves, and good ventilation. Dilute solutions still burn. Higher concentrations eat into the bench top, not just your experiment. Regular safety training and a culture that takes chemical handling seriously prevent a lot of accidents. That’s not just good practice, it makes sure everyone gets home in one piece.