People in the research world once saw Tetramethylammonium Nitrate as something of a curiosity. Early on, chemists tinkering with quaternary ammonium salts began isolating it in small batches, fascinated by its unique structure. By the mid-20th century, this compound started cropping up in organic synthesis labs as scientists explored its uses in non-aqueous media and ionic liquids. Industry professionals saw potential in its stability and solubility, and soon it found its way into more mainstream applications. Over the decades, incremental tweaks in process chemistry improved its purity, moving away from older, less efficient preparation routes. These historical experiments and gradual refinements shaped the modern production of Tetramethylammonium Nitrate, opening up doors to key studies in reactivity and safety.
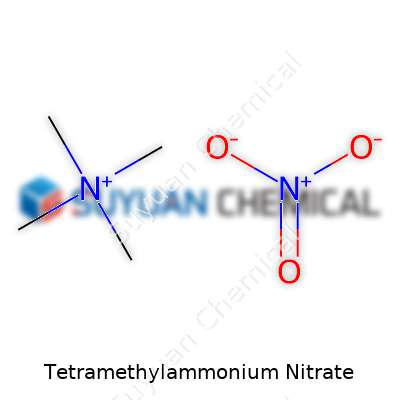
Tetramethylammonium Nitrate stands out among organic nitrates because of its structure—four methyl groups connected to a central nitrogen atom form a strongly charged cation, paired with a nitrate anion. This white, crystalline solid carries a reputation among chemists for its strong ionic character and ease of handling compared to many other nitrate salts. In lab settings, this product shows up as both a reagent and sometimes a catalyst. It feels surprisingly sturdy for a compound containing the notoriously reactive nitrate ion, which explains its traction in both synthesis and analytical chemistry. The compound’s commercial-grade samples now come with high purity, often well over 98%, intended for specialized uses rather than general commodities.
Looking at Tetramethylammonium Nitrate from a practical angle, its appearance is that of a white powder, sometimes forming crystals if prepared under the right conditions. It absorbs water from the air easily, so leaving it open on a lab bench turns it sticky. Its melting point sits just above room temperature, about 127°C, and it breaks down at higher temperatures, releasing toxic fumes. Chemically, the combination of a bulky cation with nitrate leads to decent solubility in water and many polar solvents, opening up options for use in various liquid-phase reactions. One thing that always stands out: it tends not to explode under normal lab use, unlike some other nitrates, which makes it convenient where safety matters.
Marketed Tetramethylammonium Nitrate usually gets packaged with a minimum assay over 98%, and moisture levels below 1%. The product comes in sealed, light-resistant containers, often labeled with the substance’s molecular formula (C4H12NNO3), weight, batch number, and hazard identifiers according to GHS standards. Labels emphasize toxicity and environmental risks, which reflect the standards set by agencies like OSHA and REACH. Accurate documentation accompanies every batch, including certificates of analysis and detailed safety data sheets, so end-users get a transparent look at what they’re working with.
Chemists preparing Tetramethylammonium Nitrate start by reacting Tetramethylammonium Hydroxide with a strong acid like Nitric Acid. Mixing cool, dilute solutions helps avoid excessive heat and side reactions. The process goes smoothly if you watch the temperature—the exothermic neutralization needs care to avoid runaway reactions. After getting the nitrate salt in solution, careful evaporation or cooling brings out the crystalline solid. Impurities, especially water and residual acids, get removed through successive recrystallization or drying under vacuum. This step-wise method achieves high purity fit for demanding applications, but it calls for trained hands and close monitoring because of the reagents involved.
This compound works as a reactive nitrate source for certain organic transformations. Its solubility in aprotic solvents allows unique chemistry not possible with inorganic nitrates. People have used it in alkylation, methylation, and mild oxidation reactions, sometimes benefiting from the organic solubility of the tetramethylammonium ion. In electrochemistry, it serves as an electrolyte standard, especially for non-aqueous systems. Attempting to modify its structure rarely makes sense because the cation's stability and the nitrate’s reactivity define its usefulness. Some researchers experiment with swapping the nitrate for other anions, trying to generate new ionic liquids, but even small tweaks in the cation significantly change properties and practical safety margins.
Tetramethylammonium Nitrate goes by a laundry list of names, depending on the context. Chemists may use its systematic name, N,N,N-Trimethylmethanaminium Nitrate, but most people stick with the basic Tetramethylammonium Nitrate. Shortened terms like TMAN or TMANitrate pop up in some technical literature or commercial product lists. Certain suppliers list it using catalog codes, reflecting whatever in-house system makes their logistics run smoothly. Still, the fundamental chemical structure remains constant no matter what you call it.
Handling Tetramethylammonium Nitrate requires a steady approach to safety. Safety glasses, nitrile gloves, and lab coats form the baseline gear—respect for nitrates never fades, no matter how familiar a chemist gets. Ventilation matters since excessive heat or accidental acidification can generate toxic fumes, including nitrogen oxides. Direct exposure causes irritation and, in severe cases, more systemic toxicity. Operations involving larger quantities follow strict protocols, like segregating it from strong reducers, halides, or combustible materials. Waste disposal means diluting solutions and neutralizing them under controlled conditions before sending them to approved chemical waste processors, avoiding any shortcuts that put people or the environment at risk.
Tetramethylammonium Nitrate’s main work takes place in specialist chemistry labs, from supporting organic synthesis—methylation reactions, transesterification, and selective oxidations—to acting as a phase transfer catalyst and an ion source in electroanalysis. Some researchers use it in ionic liquid research because of its well-defined ionicity and clean thermal profile, making it valuable for testing conductivity and solubility models. Analysts sometimes reach for it in high-performance liquid chromatography (HPLC) due to its stability and rapid dissociation in solution. Although some have looked for industrial-scale roles in fuel additives and explosives, safety and toxicity concerns keep those uses rare, with most technical work still happening at the bench scale.
Research around Tetramethylammonium Nitrate never stops growing, especially as new synthetic pathways demand safer or more versatile reagents. Several studies examine its structure as a prototype for other ionic liquids, seeking to balance conductivity, viscosity, and volatility for device development. Analytical chemists run tests exploring its influence on separation mechanisms, pushing for improved performance in electroanalysis. Its behavior in mixtures with other quaternary ammonium salts catches attention for building better battery electrolytes and fuel cell components. What drives much of this research is the search for alternatives to more hazardous nitrate salts, with Tetramethylammonium Nitrate as a possible step in the right direction.
Toxicology studies point out some real risks with this compound. Acute exposure to tetramethylammonium ions carries significant danger—notably, nerve toxicity. Symptoms after ingestion or substantial inhalation can turn serious, involving muscular weakness, respiratory distress, and cardiovascular complications. The nitrate part adds concerns about methemoglobinemia, especially for infants and sensitive populations. Research using in vitro and animal models pushes for lower occupational exposure limits and calls for better worker education. Spills and accidental releases can stress aquatic life in water systems, driving stricter monitoring by environmental authorities. Lab training programs push for minimum exposure and rapid response tactics in case of an accident.
Future work with Tetramethylammonium Nitrate rests on a mix of caution and promise. Chemistry teams will keep probing new uses as a model compound for green ionic solvents and more efficient electrolyte systems. Safety remains a roadblock—both in terms of chronic toxicity and rare but severe acute exposure incidents. Innovation hinges on automation in preparation and handling, aiming for closed systems that cut direct contact. Environmental regulations may push development toward derivatives with lower bioavailability or faster breakdown. Research continues to balance the push for higher-performance reagents against a firm commitment to personal and environmental safety. A future where this compound serves as both a tool for discovery and a model for best practices in chemical manufacturing feels within reach, provided teams keep channeling resources into both technical and safety-driven studies.
Tetramethylammonium nitrate doesn’t grab headlines like common chemicals. Scientists and engineers know it as a reliable compound in controlled environments. Most folks outside the lab probably won’t come across it at all. But anyone with a background in chemistry or industrial processes eventually bumps into it, usually in very specific work.
This compound stands out for its role as a reagent and as a source of nitrate ions. If you’ve ever worked in analytical chemistry, you’ve likely used it for making precise measurements. Researchers often count on it during synthesis, where getting a consistent outcome really matters. Its crystal form means it can be measured carefully and gives labs more control than using other nitrate sources.
Lots of industries have different nitrate salts on hand, like sodium or potassium nitrate. But tetramethylammonium nitrate brings a few quirks. Its unique structure means scientists can dissolve it in organic solvents more easily. Some complex chemical reactions depend on this level of solubility. The ammonium part—those four methyl groups—cuts down on water’s interference, so folks working on organic compounds get less unintended side-reaction.
Beyond the benchtop, this compound makes a name for itself in electronics manufacturing. It’s a small but important piece in the process that etches microchips. Making computer chips relies heavily on purity and precision. The wrong impurity at the wrong time ruins a batch. Tetramethylammonium nitrate dissolves cleanly and leaves few residues behind, which is one big reason it’s earned a solid spot on the production line.
In my own time handling chemicals, safety is something you never want to overlook. Tetramethylammonium nitrate falls under that rule, too. Its combination of organics and nitrate groups means it can behave dangerously if mishandled, especially if there’s any heat or a spark in the picture. People working with it need clear safety training and access to proper storage—no shortcuts allowed.
Public health agencies recognize tetramethylammonium compounds as having toxic effects on nerves and muscles at high exposure levels. Even small amounts used often mean solid housekeeping and careful waste disposal. Most workplaces stick to strict protocols, and folks in smaller operations would be wise to follow that example.
Any time you’re using a chemical with risks, it’s smart to ask if an alternative can do the job. Right now, tetramethylammonium nitrate delivers the performance needed in electronics and high-level labs. But scientists keep searching for greener or safer options, just like what happened in cleaning products or fuel additives. Until something better comes along, using it responsibly seems like the best path.
Companies and institutions have a duty to keep communities safe. That means paying attention to best practices, updating procedures when new data rolls in, and making sure workers understand both how and why certain steps are in place. Good communication, regular training, and a willingness to adapt—that’s what keeps risks low while innovation keeps moving forward.
Tetramethylammonium nitrate doesn’t show up much in daily life. In labs and some specialty industries, it fills a niche. Many wonder if it brings the same risks as other ammonium nitrates people read about in the news, usually after an accident or explosion. Here’s a real-world look at what this chemical brings to the table: it’s both hazardous and potentially explosive under certain circumstances.
By looking at older mishap records, chemists notice that tetramethylammonium complexed with nitrate can behave unpredictably. Back in 2007, a researcher at UCLA reported a rapid decomposition event linked to this substance. It wasn’t on the scale of an industrial accident, but it left a mark on policy and reinforced the message that handling this compound calls for respect and planning. Data from the National Institutes of Health and the European Chemicals Agency describe it as having both toxicity to humans and environments, along with “explosive” and “oxidizing” tendencies. Each of these warning signs points to hard-learned lessons.
Molecules like tetramethylammonium nitrate contain both fuel components (the ammonium part) and strong oxidizers (the nitrate part) within one package. That sort of molecular setup means energy can release suddenly if the wrong conditions come together—heat, friction, or mixing with the wrong chemicals. Incidents involving similar substances, like tetramethylammonium perchlorate, show that relatively small amounts can detonate in lab or plant environments if things get out of hand.
Testing this material in lab settings reveals another key feature: even a small shock or bit of static can start decomposition. This sort of instability limits its use to places with trained teams and proper containment. Story after story in chemical safety journals points to the same lessons: keep it dry, cool, sealed, and track every gram moved. I’ve seen anxiety on the faces of colleagues tasked with cleaning up even a minor spill, because risk isn’t just theoretical once you read the hazard data and remember past incidents.
Facilities that need tetramethylammonium nitrate must follow rules from both OSHA and EPA, including detailed labeling, storage in explosion-proof spaces, and regular inspections. One real threat involves contamination from other lab substances—most reports of runaway reactions stem from mixing with strong acids, heavy metals, or poor housekeeping on benches. Even the dust of this compound in the air presents a risk, both for accidental ignition and for health through inhalation.
Mock drills and honest safety briefings help most. At a research center I worked in, no one could use such chemicals without specific training. Visual reminders, like old photos of accident aftermaths, made guidelines stick. Getting everyone involved in safety checks and sharing what went wrong elsewhere closed a lot of gaps. For those only looking for the chemical’s basic effects without the risk, modern alternatives in research now often lean on less hazardous ammonium salts or use in-situ generation where feasible, cutting down on stored quantities at any one time.
Feedback from the American Chemical Society, safety training modules, and incident debriefs all point to the same habits: communicate, limit stockpiles, double-check everything, and never take shortcuts. Respect for this chemical comes from knowing its history, reading up on incident data, and insisting on best practices every day.
I remember working in a lab where the rules for chemical storage weren’t just guidelines—they were daily habits trained into muscle memory, especially when it came to materials like tetramethylammonium nitrate. This salty, white substance works as a laboratory reagent, but its risks go beyond an occasional spill. Exposure to air and moisture can make things go sideways fast. A dry atmosphere means less risk of clumping and unexpected reactions. That’s why a tightly sealed container, tucked away from humidity, stands as a non-negotiable. Clear, correct labeling, along with records of entering and removing stock, proves essential for everyone’s safety, not just the chemist on shift.
Temperature plays a major role. Tetramethylammonium nitrate breaks down more easily as things heat up, and this breakdown could release toxic fumes or even trigger an explosion. Cool, steady storage—think 2-8°C, away from sunlight and heat—is standard practice. Forget about tossing it onto a shelf near flammable solvents or acids. In labs where hazard maps are up on every door, these small steps mean the difference between a routine day and disaster cleanup.
Mixing this compound with organics or strong reducers isn't a rookie mistake; it’s an emergency waiting to happen. I’ve seen staffing shortages tempt people to cut corners, but the risk runs too high. Keeping tetramethylammonium nitrate well away from combustibles and reactive chemicals is non-negotiable, and sharing this knowledge matters. Every chemist should know their neighbors—chemical neighbors, that is.
Personal experience shows that a solid pair of nitrile gloves, a fitted lab coat, and goggles aren’t overkill. Splash incidents rarely warn you—they just happen fast, sometimes when you least expect it. Even short-term skin contact brings irritation. The fumes can sting the lungs. Proper extraction and ventilation get ignored way too often, but anyone who ever coughed through a bad reaction knows not to repeat that mistake. Shortcuts add up.
A spill kit close at hand means more than “being prepared” in theory. Absorbents, neutralizing agents, and containers for collecting waste—these should wait under the same roof, not across the building or down the hall. One sour experience with a floor spill taught everyone on my team the importance of training, not just twice a year, but ongoing, hands-on refreshers. Labeling hazardous waste and arranging proper collection matter for health and for passing regulations. Never send this down the drain—wastewater systems can’t handle it safely and neither can fish downstream.
People working with tetramethylammonium nitrate need safety data sheets, visual guides, hands-on demos, and reminders about risk, from high school chem students to experienced researchers. Nobody forgets the videos from real-life chemical accidents—fear isn’t the goal, but respect for the process comes from knowing what can go wrong. With clear communication, regular checks, and holding each other accountable, the odds tip towards safe science and long careers.
Tetramethylammonium nitrate might sound like a mouthful, but at its core, it’s a simple compound: C4H12N2O3. This formula tells us every molecule holds four carbon atoms, a dozen hydrogens, two nitrogens, and three oxygens. The molecular weight, which you get by adding the atomic weights—carbon (12.01), hydrogen (1.008), nitrogen (14.01), and oxygen (16.00)—lands around 136.15 grams per mole.
Why bring this up? I remember struggling in college chemistry, rolling my eyes every time another formula popped up. Decoding the numbers felt pointless, until a professor dropped a story about an accidental mix-up in the lab. Even a small miscalculation on a formula, he said, can wreck research or put lab staff at risk. That's real—nitrate compounds can be volatile. The details matter.
Once you know the innards of tetramethylammonium nitrate, you start to see why accuracy pays off. This substance gets used as a phase-transfer catalyst and for analytical chemistry. Getting the formula or molecular weight wrong doesn’t just mess up math on paper. It trips up every calculation that follows.
A simple slip can throw entire experiments off balance. Dose calculations go sideways. Yields don’t match up. Wasting time, money, and resources doesn’t help any scientist sleep at night. We’ve seen reports on labs recalling batches just because someone put down a wrong molecular weight on the data sheet.
Nitrate in the name hints at risk. Even small amounts of the wrong ammonium nitrate mix have caused explosions in the past, from lab mishaps to industrial accidents. Going by the numbers—between 2010 and 2020, more than a dozen documented lab mishaps involved mixing up ammonium-containing reagents, sometimes fatal. One common factor: incorrect labeling or poor record-keeping on formulas and weights.
In my own work, the margin for error felt tight. Once, our group almost ordered the wrong salt from a chemical supplier with a similar name. The only reason we caught it: double-checking the formula as policy before purchase. That helped us learn, fast, that C4H12N2O3 isn’t just trivia—it’s a safety checkpoint.
Clear labeling, ongoing training, and reliable sources help teams avoid mistakes. Anyone prepping solutions needs quick, clear access to up-to-date data: molecular weights, chemical formulas, hazard ratings. I’ve seen teams print laminated sheets with all common reagents and their numbers. Others use shared digital records that staff update regularly.
Encouraging a safety culture, where people speak up and fact-check each other, also makes a difference. Real-world chemistry isn’t just about what’s in the beaker. It’s about building habits that save time, money, and lives.
Memorizing formulas and molecular weights can feel like busywork until you’ve been there for the fallout of a small error. Relying on fact-based, reviewed information pays off. Tetramethylammonium nitrate is just one of thousands of compounds, but the lesson sticks: details shape outcomes.
Tetramethylammonium nitrate isn’t a household name, but anyone who’s spent time in a lab working with specialty chemicals knows it comes with serious hazards. This compound brings together the powerful, fast-acting nerve agent features of the tetramethylammonium ion with the oxidizing punch of nitrate. The science is clear: even small exposures can trigger severe poisoning. If you get any on your skin or breathe in its vapors, the symptoms hit early. Workers can start shivering, sweating, or feeling dizzy without much warning. So, keeping this substance under strict control isn’t just bureaucratic red tape — it’s about people getting home safe at night.
No single fix or magic product stops all hazards, but basic principles serve anyone who faces a spill. Forget gloves and goggles thrown on as an afterthought. Splash-proof suits, chemical-resistant gloves rated for organic ions, and sealed respirators should be on hand from day one. Public health evidence backs this up. The National Institute for Occupational Safety and Health (NIOSH) and Chemical Safety Board reports show that proper PPE cuts incidents in half. I remember my own early years, watching a colleague respond to a minor splash without a face shield. They landed in urgent care after a couple hours — the lesson stuck with every student around. Don't fudge the safety basics.
Ventilation proves another critical piece. Exhaust hoods with HEPA and activated carbon filters make a real difference. Even small spills can vaporize, and breathing in a few milligrams of tetramethylammonium is enough to set off acute neurological symptoms. So many facilities leave the fan off for noise reasons, but that's a risky shortcut. Proper airflow keeps everyone safer, and the difference shows up in plant health logs and staff turnover rates.
Responders should clear the area and contain the contamination. Absorbent pads made for strong oxidizers work better than generic spill kits. Keeping dry sand or vermiculite on hand can help if things spread beyond a benchtop. Once contained, material goes into sealed, clearly labeled hazardous waste containers. Skipping steps or using the wrong clean-up gear turns an easy fix into a hazmat circus. State and federal guidelines call for consulting a chemical hygiene officer before even moving the waste, and with good reason.
Medical assessment can’t be shrugged off. People exposed to even small amounts need immediate review by health professionals familiar with toxicology. Bloodwork for cholinesterase activity and close pulse monitoring mean the difference between a scary afternoon and long-term injury.
Training and safety drills have to be ongoing. Signs and written protocols do little if the organization forgets about refreshers. Teams I’ve worked with, who held quarterly walk-throughs and open safety meetings, avoided serious incidents for years — the data tracks with OSHA findings on chemical accident rates. Open communication counts, too. People more likely report near misses when they know no one will get blamed for honest mistakes.
Improving spill response for tetramethylammonium nitrate isn’t about buying new gadgets, mostly. Strong habits, tough gear, and honest reporting keep workplaces secure. That’s the kind of approach everyone deserves, whether you’re in a megacorporation lab or a small community college facility.