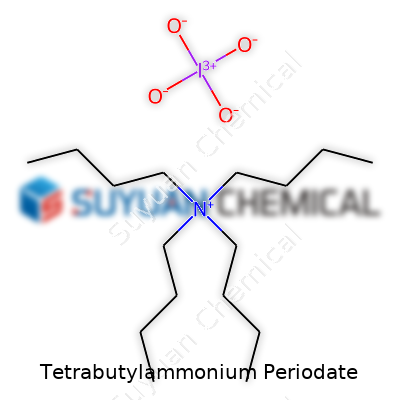
Tetrabutylammonium periodate first drew chemists' attention as the field of organic synthesis expanded during the mid-20th century. The appeal lay in pairing the oxidative power of periodate ions with the solubility and handling advantages provided by the bulky tetrabutylammonium cation. This combination helped researchers bypass solubility limitations common to traditional sodium or potassium periodate in non-polar solvents. With this salt, nearly every synthetic laboratory working on glycol cleavage, oxidative polymer modification, or late-stage functional group transformation gained an easier route. Early literature, including a 1967 publication detailing its isolation and reactivity, opened doors for broader usage. Chemists who handled many classic oxidants, from permanganates to chromates, suddenly found a mild and selective partner that avoided the environmental headaches of heavy metals.
In bottles labeled for the experienced synthetic lab, tetrabutylammonium periodate typically arrives as a white crystalline solid, sometimes clumped from slight hygroscopicity. This salt helps bridge aqueous and organic chemistry, holding both a large organic cation and a powerful IO4- anion. The material owes its popularity to reliable purity, repeatable reactivity, and firm documentation for both safety and handling. This salt often appears under synonyms such as “TBA periodate” or “Bu4NIO4”. CAS numbers on certificates of analysis let researchers confirm they’re getting the right compound for applications ranging from oxidation to analytical chemistry. With growing demand for selective and milder oxidation techniques, the role of this versatile reagent continues to expand.
Anyone who has worked in a synthesis laboratory over several winters knows the frustration of insoluble oxidants. Tetrabutylammonium periodate brings the IO4- anion into solution with surprising ease, dissolving in many organic solvents—from acetonitrile and dichloromethane to slurries in alcohol. The salt’s melting point hovers near 130°C, although decomposition can start lower if heated too rapidly. White or off-white in color, it forms needle-like crystals or can be milled into a fine powder for faster dissolution. Standard analytical work, such as elemental analysis or NMR, confirms its structure and purity. Periodate itself remains a strong oxidizer, and the tetrabutylammonium partner enhances access to delicate organic substrates that typical aqueous oxidations might ruin.
Labs using tetrabutylammonium periodate expect manufacturers to certify its minimum purity (usually 97% or greater), absence of other halate or tetraalkylammonium impurities, and batch-to-batch consistency. The labeling must emphasize its oxidizing nature, as this guides storage decisions and safety procurement. Good vendors provide certificates of analysis, outline recommended storage conditions (cool, dry, dark), and suggest shelf-lives. If handled incorrectly, the material can degrade or cake, raising doubts about reactivity and consistency. Large research institutions require comprehensive safety data sheets (SDS) and clear documentation about transportation hazards, and this chemical’s packaging reflects such regulatory scrutiny.
Most lab-scale syntheses start with simple reactants: sodium periodate and tetrabutylammonium bromide. By metathesis in a suitable solvent, such as water followed by extraction with organic solvents, the tetrabutylammonium periodate separates out. Filtration removes sodium bromide, and careful evaporation yields the crystalline product. In some settings, extra washing with cold solvents like acetone helps purge any last traces of inorganic salts. This approach yields high purity material without special equipment, and the method appears in standard synthetic chemistry references. Some industrial suppliers may modify parameters to boost efficiency, control particle size, or tailor purity for large-scale users. For researchers, the ability to prepare this salt on a modest bench setup lowers costs and gives flexibility to generate needed quantities on demand.
A veteran in the field can recall the days before selective oxidative cleavage was routine. The periodate ion cleaves vicinal diols with surgical precision, giving two aldehyde or ketone fragments. The tetrabutylammonium form amplifies this reactivity by transferring periodate into organic media, allowing oxidations in solvents like acetone and dichloromethane. Standard applications extend to glycol cleavage in carbohydrate chemistry, modification of lipid and sterol structures, and functionalization of sensitive organics that decompose in aqueous solutions. The salt can take part in further ionic exchange to introduce alternative cations, but its main influence lies in unlocking periodate’s selective power for modern synthetic schemes. In polymer labs, the salt facilitates modification of polysaccharides for hydrogels and controlled drug release, thanks to its mild yet effective oxidizing nature.
Researchers working across disciplines may call this compound by several names, depending on their background. “Tetrabutylammonium periodate” stands as the most descriptive for chemical ordering. Informal shorthand often appears as “TBA periodate” or “Bu4NIO4,” reflecting its ionic components. Published work from organic, polymer, or analytical chemistry sometimes uses names referencing its use: “organic-soluble periodate” or “lipophilic periodate salt.” Product catalogs maintain clarity with entries bearing the CAS Registry Number 62778-11-4. Unambiguous identification keeps research results consistent, avoids confusion during procurement, and standardizes lab procedures despite regional or application-specific jargon.
Over years in the lab, I have learned that underestimating oxidizers never ends well. Tetrabutylammonium periodate requires gloves, eye protection, and a well-ventilated working area. Safety data clearly mark it as harmful if swallowed and irritating to eyes and skin, and inhaling dust carries respiratory risks. The oxidizing potential urges special separation from combustible materials and reducing agents. Routine audits in regulated labs demand secondary containment, locked storage, and clear signals for emergency responders. Industry standards require updated training, so even those moving a bottle across the lab understand the risks of static discharge and accidental spills. Waste streams including this reagent move under hazardous materials protocols. These habits keep personnel, equipment, and the environment safer, even as research grows more ambitious.
Chemical innovation often springs from simple solutions to persistent problems. Tetrabutylammonium periodate solves the issue of periodate’s limited solubility in non-aqueous systems, making it valuable in organic synthesis. Carbohydrate chemists depend on this reagent for oxidative cleavage of sugars, generating smaller building blocks vital to drug development and diagnostics. Polymer scientists turn to it for selective oxidation of polysaccharide backbones, enabling surface functionalization or hydrogel formation. Lipid analysts use it for double bond localization, and environmental labs see value in its precise oxidation of certain pollutants. Because it functions cleanly in both academic and industrial projects, this salt finds roles from pharmaceuticals and materials science to specialty coatings and analytical reference standards.
Research on tetrabutylammonium periodate accelerates as new applications for selective oxidation appear each year. Laboratories working with complex biomolecules seek milder, more controllable oxidants, turning to this salt for carbohydrate and glycoprotein modification. Polymer R&D employs periodate to craft hydrogels responsive to pH, light, or mechanical stress. Analytical chemists use the salt in sensors, microfluidic devices, and as a benchmark for validating oxidative capacity in process monitoring. As sustainable chemistry grows in importance, researchers investigate greener synthesis routes for this reagent, reducing waste and by-products. The spread of this salt across so many research fields stands as evidence for its adaptability and enduring scientific value.
Safety officers always ask about toxicity when introducing a new reagent. Researchers determined that periodate salts, while powerful oxidizers, exhibit lower acute toxicity than heavier metal-based oxidants long used in similar applications. Animal studies show effects on red blood cells and kidneys at high doses, but typical laboratory handling under recommended procedures avoids these risks. Chronic exposure remains unstudied at scale, so institutions recommend engineering controls to cut down dust and inhalation hazards. Regulatory groups such as OSHA and the European Chemicals Agency set permissible exposure limits and handling guidance to minimize accidents. Responsible use demands respect for both the chemical’s capabilities and its risks, balancing laboratory needs with staff well-being.
Demand for chemoselective, sustainable oxidations only grows as pharmaceuticals, materials, and biotechnology look to improve efficiency and lower environmental burden. Tetrabutylammonium periodate sits at a crossroads, offering organic solubility, operational simplicity, and environmental advantages over legacy metal oxidants. Moving forward, focus centers on greener synthesis, improved waste management, and enhanced selectivity. Higher-throughput screening, process automation, and integration into continuous manufacturing likely expand the salt’s role. If regulatory and supply chain frameworks evolve alongside technical innovation, tetrabutylammonium periodate will play a central role in reshaping synthetic chemistry over the coming decades. Researchers who keep this reagent at hand stand ready to build cleaner, smarter reactions for a wide range of modern industries.
Tetrabutylammonium periodate brings something powerful to the chemistry table. As a reagent, it works like a tool for chemists who want to transform one molecule into another. This compound isn’t just some obscure lab ingredient. It pops up in surprising places, especially where clever and efficient oxidation reactions matter. The big draw? It’s an oxidizing agent that’s strong but straightforward enough for both research and practical jobs.
Out in the world of organic chemistry, innovation often comes down to finding the right kit to build or break apart molecules. Tetrabutylammonium periodate steps in when old-school reagents like sodium periodate fall short, especially in environments where you need a non-water-based setting. Researchers lean on this reagent to split glycols into aldehydes or ketones cleanly. It remains a go-to when working with molecules that water can mess up. In my university lab days, the difference in ease of work-up between traditional periodate and this “greener” version saved hours of hassle. Reactions run more smoothly, and the results often speak for themselves.
Lab techs and analysts aren’t left out here. Tetrabutylammonium periodate finds plenty of work in testing and quality control. It helps break down complex mixtures so detectors can spot target molecules more easily. Hospitals, universities, and government labs all rely on these kinds of chemical shortcuts to speed up analysis. It becomes particularly relevant in situations where separating and identifying small differences among similar compounds matters. Based on what I’ve seen, swapping out harsher or more toxic reagents for tetrabutylammonium periodate can also help laboratories stay within environmental safety guidelines.
Chemistry has been under pressure to clean up its act. Tetrabutylammonium periodate takes part in this by offering alternatives that are a bit gentler on the planet. By working in solvents other than water and sometimes under milder conditions, this compound often makes reactions less wasteful and easier to work up. For chemists who grew tired of fighting with endless extractions and separations, this means less solvent use and a better shot at recycling materials. With green chemistry grant money flowing, research groups keep using and studying periodate options to push for lower-impact processes. That change in mindset is real; it shifts chemistry from being “big and dirty” to more nimble and clean.
Of course, no chemical comes free of trade-offs. Periodates carry their share of safety warnings. They can be powerful oxidizers, and anyone working with them needs proper training and equipment. In my experience, thorough planning and clear protocols prevent most accidents. Factoring in environmental impact, some companies now blend safer working conditions with new containment tech. New product packaging already reduces spill risks and improves shelf life. The big leap forward might come from better education—making sure professional chemists and students focus on hazard reduction and responsible disposal.
Innovation doesn’t stand still. As industry’s push for greener and more efficient tools gathers steam, tetrabutylammonium periodate keeps showing up as a top pick. Its role in simplifying tricky transformations, improving lab safety, and supporting sustainable chemistry all matter. Based on what I see in grants, journal articles, and real experiments, people aren’t finished pushing the boundaries of what this versatile reagent can do.
Tetrabutylammonium periodate isn’t the sort of chemical that pops up in small talk, but it gets plenty of attention in university research and specialty labs. Its formula—N(C4H9)4IO4—brings together the sizable tetrabutylammonium cation with periodate anion. Each tetrabutylammonium group chains four butyl arms to a nitrogen. The periodate works as a potent oxidizer, making this salt valuable far beyond simple mixing or diluting experiments.
Molecular weight isn’t just a number in a table. It shapes how chemists think about amounts, solubility, storage, and safety. The weighted sum for this compound tallies up to around 451.36 grams per mole. That figure flips from trivia to necessity once you’re standing in a lab preparing reagents, tracking inventory, or ensuring precise reactions. Over-measure or under-measure by a few milligrams and the impact ripples through an experiment’s outcome or a synthesis’s quality.
Specialty oxidants like tetrabutylammonium periodate step up when classic periodate salts don’t dissolve so well in organic solutions. Chemists run into these hurdles working on advanced syntheses—organic, medicinal, or polymer chemistry. In my graduate days, I saw the benefit of salts with large, “greasy” cations. The organic solvents we worked with wouldn’t touch simple sodium periodate, but add a tetrabutylammonium shell and suddenly, the reactions run smoother and faster. People who care about greener syntheses or fewer toxic byproducts often search for salts that sit in that sweet spot between power and safety. This salt gives that little boost without needing metal-based oxidants.
Tetrabutylammonium periodate comes up when folks push limits—like synthesizing complex pharmaceuticals or creating new functional materials. Errors in its identity or mass set off a domino effect. Lab mistakes aren’t just inconvenient—they chew up resources, time, and sometimes turn into safety incidents. Calling out the right chemical formula and knowing the precise molecular weight shortens the distance between a sharp idea and a meaningful result. No one wants to troubleshoot reactions for days, then realize the wrong salt or the wrong mass sabotaged the data.
Quality suppliers publish information openly. That transparency has saved my teams more than once. Not all suppliers keep their certificates of analysis up to date; I’ve learned to compare calculated molecular weights against certificates and labels every time. Younger scientists might shrug and trust the bottle’s claim—but catching even one error justifies the double-check. Quality research grows from these habits, not shortcuts or guesswork.
There’s a lesson tucked inside the details of tetrabutylammonium periodate. It reminds researchers never to gloss over basics. Publishing or sharing data without confirming chemical identities and weights? That undermines trust. It slows progress. This isn’t fussy bureaucracy—it’s how science builds advances people can trust, whether that’s a new medicine or a cleaner technology. Tuning into small but crucial details, like formulas and molecular weights, keeps research honest and competitive. If science ignores these, no breakthrough holds up long. Respecting the details, even in the fine print of a salt’s label, is what gives good work its backbone.
At the bench or in the lab, few topics spark as many heated arguments or wry jokes as that cabinet full of odd, finicky chemicals. Tetrabutylammonium periodate belongs to that crowd. It doesn’t scream for attention, but getting storage wrong invites accidents that ruin a research day or send folks to the emergency room. This compound, featuring both an organic ammonium ion and the reactive periodate anion, earns respect precisely because it manages to be both useful and tricky.
Certain chemicals just come with strings attached. Tetrabutylammonium periodate demands care because periodates work as aggressive oxidizers. Spilling them or letting them get cozy with organics, acids, or reducing agents can spike the risk of fire and even explosions. As a fellow chemist who’s watched a seemingly quiet lab turn frantic when someone stores an oxidizer next to solvents, I’d argue the real risk isn’t theoretical.
Temperature tells part of the story. Periodates break down if the room gets too hot, and Tetrabutylammonium Periodate doesn’t sidestep this problem. Standard practice keeps it in a cool, dry spot away from sunlight. Moisture or lingering vapors can spell trouble. Think of those times a humidity spike ruined a reagent—periodate salts take that kind of abuse poorly, sometimes forming hazardous oxygen-rich residues.
Glass bottles with tightly fitting screw caps stand out as a safe choice. Polyethylene bottles work too, so long as the cap keeps air and humidity out. Skip metal containers; periodates corrode them fast. In my own lab, we label every oxidizer with bold “Oxidizer” stickers, not just to satisfy a regulation, but to keep that bottle from landing in the wrong bin or shelf.
Real safety means distance. The best chemists treat the storage room like a chessboard. Acids, reducing agents, flammables, and oxidizers each deserve their own corner. Segregated storage wins the day. To support that claim, studies from the CSB and the Royal Society of Chemistry highlight that mishaps drop sharply when labs separate reactive chemicals. I’ve seen that myself: organized shelves mean fewer mistakes and less panic when someone can’t recall where that new shipment landed.
Security isn’t just a label on a door. Keeping a running inventory, spot-checking containers for leaks or color changes, and rotating stock (first in, first out) mean fewer surprises. I’ve seen seasoned techs catch a degraded salt before it caused a problem—just because they take five minutes every Friday to check labels and seals. If you let periodate salts sit in a forgotten corner, decomposition can turn the material dangerous and create disposal headaches.
Better signage, strict separation, and simple checklists pay dividends. In my lab, we print quick guides for new staff to follow, and that step alone has cut down on “mystery bottle” scares. Investing in silica gel packs to line cabinets helps dry the air and prevent slow moisture damage. Supplying clear plastic bins for oxidizers creates a physical barrier to spreading spills, saving time and nerves when things slip out of hand.
Treat Tetrabutylammonium Periodate as a respected guest—not to be feared, but not to be ignored. Simple precautions, teamwork, and a dose of vigilance keep the lab humming, spills contained, and research on track. Science can be unforgiving, and so can oxidizers. The best labs, and the best teams, learn this lesson before the hard way comes knocking.
Tetrabutylammonium periodate holds real power in the lab, especially for those running oxidation reactions or looking for a strong oxidizer. What’s also real is the risk: this chemical can threaten your health and safety if you don’t show it respect. Anyone who works with hazardous chemicals knows the basics—gloves, goggles, maybe a lab coat. But for some compounds, including this one, safety isn’t just a checklist. Understanding the specific dangers makes you more likely to avoid a trip to the emergency room.
Tetrabutylammonium periodate is an oxidizer, and strong oxidizers love to cause unexpected trouble. Spilled on your skin, it can burn and irritate. Breathed in, the dust or fumes attack your breathing and hit your lungs hard. I’ve seen more than one student learn this the hard way, thinking a quick sniff couldn't hurt. It can.
Even solid chunks, left lying on a bench, don’t stay safe. They react with many solvents, especially those with strong reducing abilities. Add it by accident to acetone, ethanol, or other common organics, and suddenly you’ve got heat, fumes, or even fire. Using Tetrabutylammonium periodate responsibly means knowing what it might touch and keeping incompatible substances far away.
Regular nitrile gloves and splash goggles provide a first barrier. But for longer handling—especially if weighing out many grams or transferring to solution—you’ll want a face shield or chemical-resistant apron. Chemical burns on hands or forearms aren’t just uncomfortable; sometimes, they linger. In my experience, cheap gloves can leave you thinking you’re protected, right until the tiniest splash slips underneath.
Work in a well-ventilated fume hood. The temptation to cut corners—just pipet out a small amount in the open lab—often comes back to bite. I once saw a friend race to the eyewash station after a few drops splashed from a supposedly “safe” bench. Don’t let hurried hands push safety out the door.
Store Tetrabutylammonium periodate in a cool, dry spot away from incompatible chemicals like organics and strong acids. Use sealed containers, labeled clearly with hazard warnings. Avoid glass jars with ground glass stoppers, since the salt sometimes crusts up and effectively glues the lid shut. Plastic or PTFE containers with tight screw tops stay safer, especially if there’s any chance of water condensation or humidity.
Never pipet by mouth. Automatic pipetters and spatulas provide a hands-off approach for measuring powder or solutions. Always add periodate slowly to reaction mixtures; dumping it in quickly can start off a violent reaction.
Small spills usually call for a generous scoop of inert absorbent—think sand, vermiculite, or even standard spill pillows. Don’t sweep or blow powder around, since anything airborne could end up in your eyes or lungs. For larger spills, clear the area and call the safety officer. Periodate waste must go in designated hazardous waste containers. Pour it down the drain or toss in the trash, and you risk contaminating the water supply or causing nasty reactions downstream.
If exposure happens, wash affected skin or eyes straight away with copious water. Remove contaminated clothing. Breathing in fumes or dust? Fresh air comes first and a call to medical staff follows. Your supervisor or lab manager has procedures lined up for almost every scenario—don’t feel embarrassed to sound the alarm.
Chemistry moves faster and results grow more reliable in a group that values safety. Sharing stories, double-checking your neighbor’s PPE, and never letting anyone work alone with hazardous materials are things that make a difference. Most accidents in labs I’ve seen started not with carelessness, but with overconfidence right before a shortcut. Slow down, ask questions, and look out for others. The learning sticks longer than the scars.
Tetrabutylammonium periodate stands out for its use in organic chemistry, especially when it comes to oxidation reactions. Researchers and lab techs usually reach for this salt if they want to carry out a selective oxidative cleavage or manipulate sensitive molecules. That means people keep running into the same question: does it dissolve well in water or is it more at home in organic solvents?
With simple ions like sodium chloride, tossing crystals into water gives you fairly quick results, but things grow a little unpredictable with tetrabutylammonium periodate. From my own time prepping samples, this salt tends to resist full dissolution in water. The tetrabutylammonium cation’s four butyl chains make the salt bulky, hydrophobic, and stubborn about interacting with polar molecules. People have reported only partial solubility or slow dissolution, leading to cloudy mixtures or undissolved solids. A 2011 publication in "Synthesis" journal explains that the periodate anion on its own shows good water solubility, but the tetrabutylammonium partner changes the game.
If you try to hot-stir or use ultrasonic baths, patience pays off, but you rarely get that clean, transparent solution people expect from simpler salts. In practice, researchers sometimes suspend the salt or use just enough water to help it along during reactions, then rely on mechanical stirring.
Take the same salt and put it into an organic solvent like acetonitrile or dichloromethane. Suddenly, solubility improves. The broad, greasy tetrabutylammonium cation binds more comfortably to solvents with large hydrocarbon portions. This makes sense; like dissolves like. Christodoulou and colleagues described, in a 2007 issue of "Organic Letters," that this salt dissolves reasonably well in polar aprotic solvents. For anything involving two-phase oxidations or reactions where water would be a burden, using an organic solvent often gets better results.
On top of that, the use of organic solvents expands the toolset available to chemists. Extraction, purification, and certain catalysis steps need organic compatibility, especially as more synthetic reactions move away from water as a default medium.
Plenty of students and newer lab staff wonder why people spend so much time talking about solubility. The truth is, the choice affects reaction rate, yield, and even safety. Incomplete dissolution can mean uneven reactivity and wasted starting materials. Someone once lost an entire day to a bottle of tetrabutylammonium periodate that just refused to mix until we switched solvents.
Environmental and cost concerns play a role, too. Labs lean toward solvents that allow for easier workup and lower risk. Water is cheap, non-flammable, and easy to dispose of when clean, but poor solubility pushes more users toward organic chemicals that require fume hoods, waste disposal, and careful storage.
To work with tetrabutylammonium periodate, pick your solvent with a plan in mind. If a process absolutely needs water, consider increasing temperature or agitation, but always keep safety in focus—periodates pack a punch as oxidizers. A few companies now offer pre-made solutions in various solvents, which saves time but raises costs. Others have suggested tweaking the counterion, using alternatives like sodium or potassium periodate if possible, or running biphasic systems where the salt bridges water and organic layers.
At the end of the day, matching the solvent to the salt's character keeps workflows smoother and reactions more reliable. It’s one more example of why a little chemical intuition can save hours of trial and error.