Back in the decades when organic synthesis was spreading new wings, scientists hunted for reliable phase-transfer catalysts and ion-pairing agents that could get stubborn reactions off the ground. Tetrabutylammonium hydrogen sulfate came out of that search. Chemists started using quaternary ammonium salts in the 20th century to push reactions between oil-soluble and water-soluble reactants. Tetrabutylammonium derivatives found their spot in labs because they could slip between phases and nudge things along, breaking barriers that often slowed down multi-phase chemistry. Over the years, lab experiments grew into bulk manufacturing as industrial processes required clean and manageable auxiliary reagents. Older chemists I’ve worked with still talk about the dramatic jump in yields and process reliability once salts like tetrabutylammonium hydrogen sulfate replaced trickier, less stable agents.
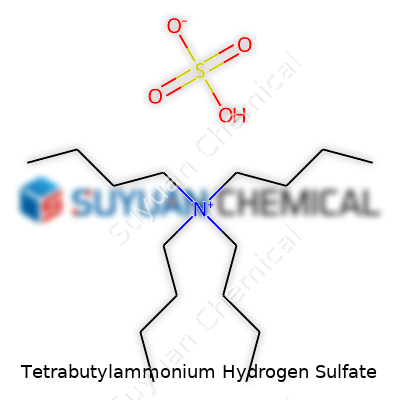
Tetrabutylammonium hydrogen sulfate, commonly called TBAHS, doesn’t grab attention in casual chemistry chats, but anyone involved with phase-transfer reactions has handled its white, crystalline form. Labs order it for its handy dual nature: one part big organic cation (tetrabutylammonium), one part acidic anion (hydrogen sulfate). Commercial bottles show up under names like N,N,N-tributylbutan-1-aminium hydrogen sulfate and TBAHS; the CAS number often provides quick assurance of authenticity for those double-checking inventory.
This salt stands up as a typical, solid quaternary ammonium compound. At room temperature, it appears as white crystals or a powder with a faint smell. In the palm, its texture has enough grit to remind me of ordinary table salt, though it dissolves more easily in polar organic solvents and water. Its melting point hovers between 190 and 200°C, making it stable enough for routine lab work without much risk of decomposition at standard reaction temperatures. The molecule’s four butyl groups tag onto a central nitrogen, wrapping up the hydrogen sulfate as a tight ion pair. In my experience, it’s non-volatile and doesn’t bring any unpleasant fumes to the bench—one less thing to worry about compared to volatile acids.
Suppliers usually list purity, water content, and trace impurities right on the label. Any container that doesn’t clearly label these fails to meet best practices, since accurate information keeps reactions predictable. Research-grade TBAHS often comes at 98% or higher purity, although for tougher industrial needs, extra processing can push that higher. Packaging touches on safety too: airtight containers keep out moisture, as quaternary ammonium salts can pull water from humid air and clump up over time. Proper chemical labeling with hazard pictograms, lot numbers, and expiration dates helps avoid confusion and mix-ups, especially in labs with lots of hands on deck.
A reliable way to make TBAHS uses tetrabutylammonium bromide or chloride and a source of sulfuric acid. Mixing a solution of tetrabutylammonium salt with sulfuric acid leads to ion exchange. The hydrogen sulfate swaps in for bromide or chloride, and the insoluble byproduct (like sodium chloride if using tetrabutylammonium chloride and sodium hydrogen sulfate) can be filtered out. After concentration, the resulting TBAHS can be re-crystallized from solvents such as ethyl acetate or ethanol to clean out any unreacted ingredients. I’ve watched this done in both teaching labs and pilot plant scales—a straightforward prep that rarely surprises if stoichiometry and cleanliness are respected.
What makes this salt valuable isn’t so much its direct reactivity but the way it shuffles ions between layers. In reactions where aqueous and organic phases sit on top of each other, TBAHS ferries charged species (like hydroxide or halides) across, letting things react that wouldn’t otherwise meet. Alkylation, epoxidation, and oxidation reactions run smoother and faster with TBAHS added—I've watched sluggish conversions leap to completion thanks to the phase-transfer boost. It plays well with a raft of nucleophiles and electrophiles. Modifying TBAHS—say, swapping the hydrogen sulfate for another acid anion—creates a family of analogues, each with its own quirks for different reactions.
Depending on which supplier or catalog you pick up, this compound goes by several names. Tetrabutylammonium hydrogen sulfate is the formal chemistry name, but catalogs often abbreviate it to TBAHS or list it as N,N,N-tributylbutan-1-aminium hydrogen sulfate. Other listings sometimes call it Tetrabutylammonium bisulfate or Tetra-n-butylammonium hydrogen sulfate, though the core structure stays unchanged. The name matters if you’re combing through literature or trying to order the right stuff for a new project; different countries and manufacturers sometimes slip in minor naming tweaks, but the CAS number (32503-27-8) usually provides a quick confirmation.
While experience tells me TBAHS behaves in a predictable, manageable way, chemical safety still matters. Skin and eye contact brings irritation, so standard gloves, goggles, and lab coats stay on. The MSDS for this material warns against inhaling dust, though it doesn’t spread through the air easily in normal use. Spills clean up without fuss using water and soap, but good housekeeping cuts down on cross-contamination risks. In the waste stream, TBAHS should be collected with other organic laboratory waste, making sure not to dump it into regular drains; the ammonium ion and sulfate content can pose an environmental hazard in high enough concentrations. Operating with clean, labeled bottles and storing away from acids or oxidizers extends product life and keeps workplace incidents in check.
The biggest impact for TBAHS falls in organic synthesis—especially in multi-phase reactions where traditional bases or acids move sluggishly. Epoxidations, oxidations using permanganate or hypochlorite, and nucleophilic substitutions all benefit from a well-chosen phase-transfer catalyst like TBAHS. In industries preparing fine chemicals, pharmaceuticals, or specialty polymers, this salt stands out for its reliability. When working on drug intermediates or specialty monomers for new polymers, using TBAHS simplifies the process, often dropping the reaction time and reducing the need for harsh conditions. Water treatment, analytical chemistry, and electrochemistry also tap into its ability to shuttle ions effectively, though the bulk of revenue seems to come from organic labs and chemical manufacturing.
Research on TBAHS keeps churning out tweaks and applications. Some chemists look at replacing TBAHS with greener alternatives, but most agree its combination of performance, availability, and safety is tough to beat. In green chemistry, newer solvents and cleaner production lines have sparked interest in reusing or recycling TBAHS efficiently. Other studies dig into the salt’s behavior in non-aqueous systems, trying to expand its reach into battery chemistry or nano-material synthesis. My colleagues testing emerging catalytic systems often count TBAHS as a dependable benchmark, seeing how new compounds measure up against a tried-and-true performer.
TBAHS, like most quaternary ammonium salts, demands caution in handling despite a relatively benign reputation in chemical circles. Acute toxicity sits low compared to heavy metals or volatile organics; most studies show low risk in controlled exposures. Chronic effects aren’t as well mapped out, though; persistent skin or eye contact, or environmental build-up over time, brings potential issues. Studies in aquatic systems show that larger spills could affect lipid membranes in small organisms. Responsible handling means keeping exposure down, properly managing waste, and never getting comfortable or careless with protocols, especially as new regulatory focus lands on quaternary ammonium compounds in waste streams.
Looking ahead, TBAHS stands a good chance of staying on bench tops and production sites, even as green chemistry nudges folks toward less persistent salts in some settings. Producers respond to regulatory and end-user demand by fine-tuning purity and packaging, offering more environmentally friendly supply chains, and engineering ways to recover and reuse spent catalyst. Researchers trial modifications to the basic tetrabutylammonium structure, trying to match performance with easier biodegradability. Battery chemistry, ionic liquids, and smart drug synthesis keep opening new doors for salts like TBAHS; the hunger for more efficient, selective, and sustainable chemistry keeps sharpening its place in the world. In all this, nothing quite replaces hands-on experience working with the salt—good technique, steady protocols, and real-world know-how keep it useful and safe in both old and new applications.
Curiosity about tetrabutylammonium hydrogen sulfate usually pops up in labs, industrial workshops, and universities. You see it on supply shelves labeled as a phase transfer catalyst, but the story stretches a bit further. Tetrabutylammonium hydrogen sulfate doesn’t just sit in bottles gathering dust; researchers and chemists lean on it to push tricky chemical reactions over the finish line. Getting polar and nonpolar compounds to mingle isn’t simple. This is where tetrabutylammonium salts lend a hand and help transfer ions from one phase to another, making reactions possible that otherwise stall out.
During undergraduate lab work, handling this compound made me realize just how much chemistry relies on teamwork—between molecules, solvents, and catalysts. With organic synthesis, you might be dealing with a stubborn molecule stuck in water that refuses to shake hands with an oil-based partner. Tetrabutylammonium hydrogen sulfate steps in and nudges reactants toward each other, bridging the gap that water and oil can’t cross alone. This small but crucial push often leads to shorter reaction times and less need for harsh conditions.
Real progress shows up in efficiency. Instead of wasting hours or fiddling with higher temperatures, researchers can streamline methods. For example, in my academic experience, working with quinine derivatives sped up once tetrabutylammonium hydrogen sulfate entered the mix. The product yield improved, and repeatability soared. These improvements aren’t just about convenience; they reduce energy consumption, lower waste, and even improve safety—three things that matter in both commercial settings and smaller research labs.
The reach extends past education and bench chemistry. In pharmaceuticals, making key ingredients faster and with fewer contaminants leads directly to savings on cost and environmental impact. One case in point involves the synthesis of active pharmaceutical ingredients using phase transfer catalysis—a process that now stands as a pillar in the chemical industry. Tetrabutylammonium hydrogen sulfate helps drive these reactions, offering cleaner profiles and fewer harmful byproducts.
Environmental analysis also finds a use for this compound. Extraction of pollutants from water samples often relies on phase transfer agents. When local authorities need accurate results—say, to check for illegal discharges in drinking water supplies—tetrabutylammonium hydrogen sulfate proves essential for getting clear readings. As a person involved in water testing projects, I witnessed firsthand how this chemical improved detection levels, which impacted not just the local environment but also public health decisions down the line.
Handling and disposal raise challenges. Tetrabutylammonium salts, if not managed correctly, can impact aquatic ecosystems. Factories that use these compounds sometimes struggle to treat their waste streams, and regulations are getting stricter. Findings from the European Chemicals Agency stress the need for closed systems or effective waste management in industrial settings. In academic labs, training on safe handling and disposal cuts down on accidents and contamination. I always preferred working under supervision, learning best practices from mentors who prioritized both results and responsibility.
Looking ahead, alternatives to tetrabutylammonium hydrogen sulfate are being developed, especially in green chemistry. Some researchers focus on biodegradable phase transfer catalysts. Switching to greener options could take time, investment, and proper education. Sharing practical tips and stronger cooperation between academia and industry will help this transition.
Understanding chemicals sometimes gets tangled up in jargon and hard-to-pronounce names. Tetrabutylammonium hydrogen sulfate is one of those mouthfuls, but it shows up in real labs, not just textbooks. At a glance, its chemical formula is (C4H9)4NHSO4. That string of letters and numbers reveals a lot about how this compound acts and why chemists and engineers turn to it for specific tasks.
The core of this molecule is the tetrabutylammonium ion. You get four butyl groups—meaning four whips of carbon and hydrogen—hooked up to a nitrogen atom. Toss in a hydrogen sulfate ion, and you’ve got a salt that does what many simple salts can’t. The whole molecular assembly usually appears as a colorless or white solid, eager to dissolve in water or certain organic solvents.
Tetrabutylammonium (N(C4H9)4+) comes with serious heft compared to, say, sodium or potassium ions. These big, flexible organic arms help tug molecules from one phase to another, which proves very handy in industry and labs.
Take it from someone who has watched a reaction grind to a halt without the right catalyst or salt in the mix. Compounds like tetrabutylammonium hydrogen sulfate act as phase transfer catalysts. They boost reaction speed by ferrying ions between oil and water layers—without them, some processes crawl or don’t go anywhere at all. The classic “two doesn’t mix” scenario in a beaker changes completely with this salt sprinkled in.
In organic synthesis, this chemical turns up in the preparation of a dizzying range of drugs and specialty chemicals. Companies lean on it when other salts can’t quite hack separation and purification as efficiently. Here’s another big job: environmental labs often use it during extraction procedures to pull out specific ions or contaminants for testing and clean-up. Its structure—revealed in that formula—explains why its performance outshines smaller, simpler agents that fail to shift heavy molecules across boundaries.
Unpacking safety means reading the signals hidden in structural formulas. This salt doesn’t spark dramatic explosions or acute dangers. Still, nobody should take its gentle reputation for granted. Gloves, goggles, and ample ventilation keep labs safer. Health agencies document that some quaternary ammonium compounds cause skin or eye irritation, especially when handled carelessly over time. Using basic protective equipment and storing it in dry, well-marked containers handles most of the risk. Proper waste management closes the loop; pouring it down the drain, even in low concentrations, strains wastewater systems that aren’t always ready for specialty chemicals.
Working with any chemical starts with a core fact. For tetrabutylammonium hydrogen sulfate, the formula makes its story clear: C16H37NHSO4. Every time a chemist weighs it out, they’re counting on its proven ability to change the odds in tricky reactions, speed up extractions, and support greener clean-up strategies. Its uses grew from that chemical structure—the butyl chains, the sturdy ammonium head, and the hydrogen sulfate tail—to solve real problems. Accepting what the formula teaches gives researchers, students, and industry leaders a practical tool that doubles as a bridge between pure chemistry and hard work in the field.
Tetrabutylammonium hydrogen sulfate turns up in quite a few lab experiments and some industrial setups. Its name feels like something out of a textbook, but the realities of working with chemicals like this are very hands-on. My own time in a university chemical storeroom comes to mind—labels, warnings, gloves, about twenty rules for moving a single bottle. Back then, we relied on our instincts and training as much as on the fine print in the Material Safety Data Sheet (MSDS).
Just because a chemical shows up regularly in the lab doesn’t mean it’s harmless. Tetrabutylammonium hydrogen sulfate has some real teeth if handled carelessly. If you open the MSDS, you’ll see warnings about skin and eye irritation. Accidentally breathing it in adds its own risks. I’ve seen people skip gloves because they get used to the routine. It only takes once: a split second, and someone’s brushing their face or not noticing irritation until after the fact.
There’s another layer: this compound is a quaternary ammonium salt, and many in this group cause trouble inside the body if ingested or absorbed through skin. Inhaling its dust can lead to coughing, or even more serious breathing problems. Swallowing it can trigger abdominal pain or worse, depending on the dose. Even if you’re careful, there’s always a risk that a spill or splash leads to accidental contamination.
Long work in a lab can push people to let their guard down. Nobody wants to suit up in goggles and masks all day, every day, but that’s what’s required. With repeated contact, sensitization creeps up. Allergic reactions, increased irritation, or even more chronic health complaints get reported in settings where people didn’t take those first tingling sensations seriously. I remember my own hesitation to wear a full apron—until the first time a chemical burned through my shirt and left a mark.
Moreover, not every risk stops at the door. Disposal matters. Some labs pour compounds down the drain, thinking a little dilution offers safety. That’s risky thinking. Some quaternary ammonium compounds have shown toxicity to aquatic life, even in low concentrations. Improper disposal could hurt more than just the person in the lab.
The rules set out in Safety Data Sheets demand gloves, goggles, and fume hoods. In my experience, regular training sessions make the difference. Folks new to the job need stories and real-world context, not just paperwork. They need to see how a careless moment can sideline a promising week or even end a career. Good labeling and clear instructions help, but culture decides if anyone actually follows them.
Lab managers looking to cut down risk can start with two habits: keeping dangerous chemicals locked up and making every clean-up as important as the experiment itself. A broken bottle or a forgotten spill often leads to the worst outcomes, especially after hours. For those worried about long-term consequences, substitution sometimes offers relief. If a safer chemical can serve the same purpose, it’s probably the better choice.
I’ve leaned on trusted colleagues for the latest safety updates, and I encourage others to do the same. Nothing beats a quick conversation with someone who's already learned from experience. Anyone handling tetrabutylammonium hydrogen sulfate should keep both eyes open and never shortcut the basic steps. Safety hinges on the habits you set, long before an accident ever happens.
Tetrabutylammonium hydrogen sulfate looks like just another white solid on the laboratory shelf, though it holds a mix of power and unpredictability. This compound serves in phase-transfer catalysis and organic synthesis, so researchers and chemists encounter it regularly. It carries both an ammonium cation and a hydrogen sulfate anion, which means it draws moisture from the air and carries some risk if handled carelessly.
This chemical loves moisture. Sit it out for even a few hours in a damp lab and you’ll notice clumping, maybe even some stickiness to the touch. Over time, this dampness can break down the compound or skew the results of any reaction that relies on precise measurements. That’s not something any careful worker wants to deal with, so a sealed, airtight container makes the difference. Glass jars with tight screw tops or chemical-resistant plastic containers both work, though some labs keep everything double-bagged in zip locks, just to shut out humidity.
Keeping things stable avoids big headaches. Store tetrabutylammonium hydrogen sulfate at room temperature, away from windows, hot pipes, or direct sunlight. If it’s too warm, chemical changes creep in faster. If it’s too cold, condensation builds up each time someone opens the container. The sweet spot for most chemicals in daily lab work sits around 20-25°C, and tetrabutylammonium hydrogen sulfate fits that mould just fine.
Many labs bunch chemicals together by type, but this compound proves stubborn. It doesn’t belong anywhere near strong oxidizers or strong acids. Accidental mixing—sometimes just a spill or a shared spatula—might trigger fumes or unpredictable reactions. A dedicated section of a chemical storage cabinet lowers risk. Even a simple shelf with labelled cubbyholes helps keep things in order.
Years of picking through shared university storage rooms taught me one thing: people often reach for whatever spoon or scoop they see, and mistakes happen. Once, someone used the same spatula for this compound and then for solid acid, leading to a minor reaction and a morning spent mopping up. Color-coded tools or clearly labeled small scoops, placed right next to each compound, really do cut these mistakes down.
Clear, bold labeling on containers, with name, date opened, and hazards, makes all the difference. It stops people from guessing or just assuming what’s inside. Anyone stepping in, whether new staff or a tired researcher, can check what they’re holding and read the hazards in a split second.
Research groups could lean on digital inventory systems that alert staff when a container’s been open too long, or when old stock risks breaking down. Training sessions focusing on specific chemical risks—rather than just general lab safety—raise awareness and lower incident rates. When tight budgets make these changes tough, regular team conversations about mistakes and near-accidents keep everyone’s guard up.
Safe, proper chemical storage keeps people healthy, protects research, and cuts out expensive accidents. Small steps—airtight jars, clear labels, attention to humidity and temperature—help even casual users avoid the biggest pitfalls. My own knuckles have scraped the insides of too many corroded storage cabinets to ever brush this off as just another lab chore.
Tetrabutylammonium hydrogen sulfate, with its molecular formula C16H37NO4S, stands apart from everyday salts in the lab. It looks like a white or slightly off-white crystalline powder. The grains don’t clump together from humidity as much as many other salts do. That makes scooping and measuring more straightforward, especially during rushed afternoons in a lab setting. As a chemist who’s prepared dozens of oddball reagents, having a powder that’s easy to handle feels like a win.
One thing that comes up every time this chemical gets used is the melting point. It sits around 180–185°C. That’s higher than many organic compounds, so it doesn’t melt or break down if the lab warms up—and it survives mild heating or mixing. You often find chemicals like this in reactions that need a stable ion source. Nobody wants a reagent that degrades before the experiment even begins.
Tetrabutylammonium hydrogen sulfate dissolves really well in water and a range of organic solvents, including acetonitrile and methanol. This means it hops from polar to less polar environments with minimal fuss. In practical terms, you can use it in two-phase systems or phase-transfer catalysis, where getting ions from one liquid to another is the whole point. For chemists, that flexibility saves time, eliminates weird troubleshooting, and reduces the need for multiple specialty reagents.
The powder gives off a mild, almost unnoticeable odor, which is a relief since some ammonium salts smell pretty strong. Its texture is fine and free-flowing, never sticky. I’ve often compared it to finely-milled flour. It pours and weighs without mess, so every gram lands in the reaction flask, not drifting onto your workbench or gloves. That cuts down on waste, which always matters when working with high-purity or expensive chemicals.
Unlike some lab powders that suck moisture from the air and turn into a sludge, tetrabutylammonium hydrogen sulfate remains pretty dry. This behavior stems from the big, hydrophobic butyl groups sticking out from the ammonium core, which push away water molecules. Fewer desiccant packs, less double-checking storage containers: these small things lower the day-to-day stress in any active laboratory.
Its color stays a steady white under normal conditions, and exposure to light doesn’t turn it yellow or brown. No need to store it in amber bottles or panic over shelf life after a few hours on the benchtop. Consistent appearance helps when you’re preparing multiple solutions and want to spot contamination right away.
The way tetrabutylammonium hydrogen sulfate behaves physically touches every part of how it gets used. Stable melting point, easy handling, nice solubility, and dry storage let you run reactions with confidence. Many specialty chemicals claim to do the job, but only a few actually fit seamlessly into daily lab life. By streamlining these small details, this compound helps science run just a little smoother.